Crossover Analyses
Most large-scale clinical trials use a parallel experimental design in which randomly selected subjects are assigned to one of two or more treatment Arms. Once assigned to an Arm, each subject is given a single treatment, either the drug or drugs being tested, or the appropriate control (usually a placebo) for the duration of the study. Data are collected and subjected to between-subject analysis. Large sample sizes are usually required in these studies to account for the many sources of inter-subject variation, while still enabling accurate detection of any treatment effects.
An increasingly popular strategy for clinical trials, particularly for those involving stable, chronic conditions, involves the use of a crossover design. In this design, every subject is sequentially given all of the treatments in the study. Each treatment is administered for a defined period of time. Each subsequent treatment is preceded by a recovery, or washout, period where no treatments are administered, to allow subjects’ conditions to return to their normal states. Subjects are randomized only with respect to the order in which the different treatments are administered. Because each subject serves as his or her own control, reducing the effects of between-subject variation, and because each subject can be used multiple times, crossover studies usually require far few subjects, although for longer times, than comparable parallel studies. These benefits can often outweigh the risks (subject drop-out, changing subject condition over time, carryover of one treatment to the next due to insufficient washout, or any secondary effects), associated with this design.
While JMP Clinical was initially designed for analysis of parallel studies, numerous reports in the JMP Clinical software now also support crossover analyses.
The following sections tell you how to use JMP Clinical for crossover analyses.
Using JMP Clinical for Crossover Analysis
Using JMP Clinical for Crossover Analysis
All study information must be recorded using an ADSL data set following the ADaM standard to support multiple treatment periods.
Required Variables
The variables in ADSL required for the system to support crossover include the following:
| • | Treatment Variables TRTxxP or TRTxxA: The Planned or Actual treatment for a given treatment period xx (The TRT01P, TRT02P, for example, would be the variables used to record the planned treatment for a two-period crossover analysis). |

| • | Timing Variables (TRxxSDT or TRxxSDTM) and (TRxxEDT or TRxxEDTM): Dates or Date/Times in numeric format indicating the start and end dates respectively for each treatment period xx. |
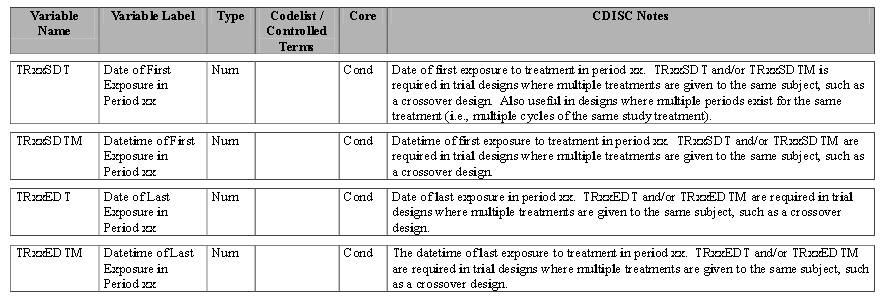
Refer to the ADaM Structure for Occurrence Data Implementation Guide v1.1 for additional information.
JMP Clinical Reports Supporting Crossover Analysis
The software supports analysis and views for crossover trials in the following reports.
| • | Adverse Event Distribution |
| • | Adverse Events Multiple Occurrences Distribution |
| • | Adverse Event Incidence Rates |
| • | Adverse Events Narratives |
| • | Demographics Distribution |
| • | Events Distribution |
| • | Events Multiple Occurrences Distribution |
| • | Interventions Distribution |
| • | Interventions Multiple Occurrences Distribution |
| • | Findings Time Trends |
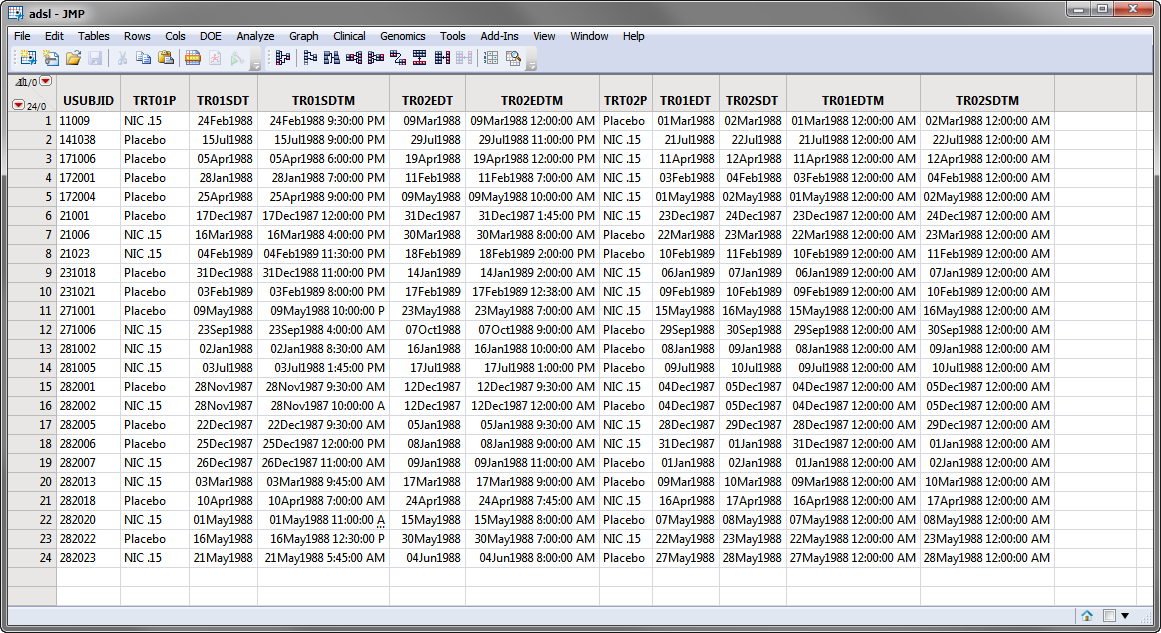
Currently crossover support is automatically detected and used if multiple TRTxxP or TRTxxA variables exist and the associated timing variables for the xx period exist and contain values. Those values might look like the values in the Nicardipine Crossover sample data set, shown below.
Note: For clarity, the majority of the adsl columns in the screen shot below above have been hidden using the Cols > Hide/Unhide command.
During the report analysis, when adsl is merged into the relevant analysis domain (for example, AE, LB, VS), the adsl treatment timing variables are compared to the timing variable in the domain and new variables: Treatment, Period, and Treatment (Period) are created. The values for these variables are assigned based on the value of the TRTxxP or TRTxxA and the value of the xx in the variable name when the start date of the domain record falls within the treatment period dates.
The SDTM data records for a subject can appear as shown in the portion of the vs domain for the Nicardipine Crossover sample data set, shown below.
Note: For clarity, some of the vs columns in the screen shot below above have been hidden using the Cols > Hide/Unhide command.
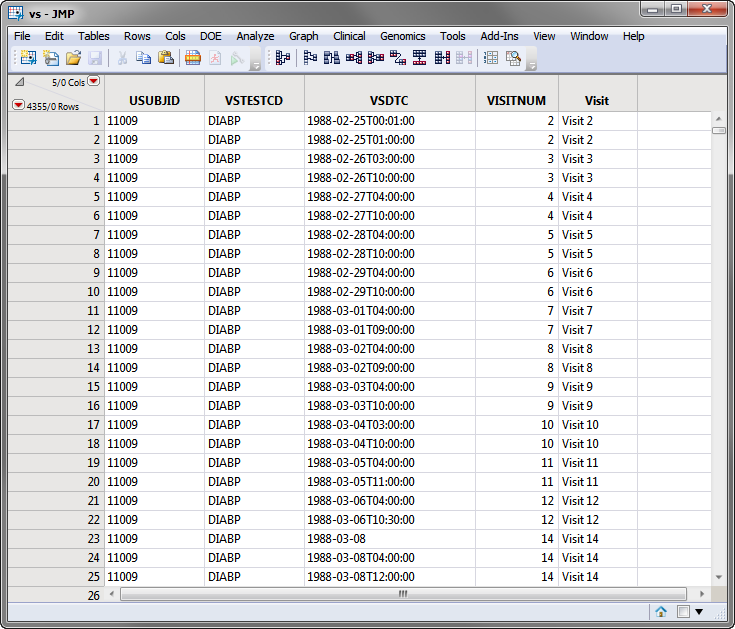
Based on comparison of the VSDTC date/time (note that SDTM follows the ISO 8601 date/time standard) with the ADSL timing date/time, the subject records are assigned to the first treatment for Visits 1-6 and the second treatment period for Visits 7-14.
Similar analyses are done for events and intervention domains. In these domains, the timing comparisons for the record is based on the start date/time. For example, with the AE domain the AESDTC variable is used to assign the treatment period; indicating that the adverse event must START within the treatment period in order to be assigned that treatment value.
Report Results with Crossover Analysis
A sample Nicardipine Crossover study (Nicardipine CO) is shipped with JMP Clinical. Refer to Nicardipine CO for information about this study.
Analysis results and report options relevant to crossover analysis are shown below.
When a crossover study is detected, the distribution results automatically categorizes adverse events by treatment period.
The results with the example Nicardipine crossover data are shown below.
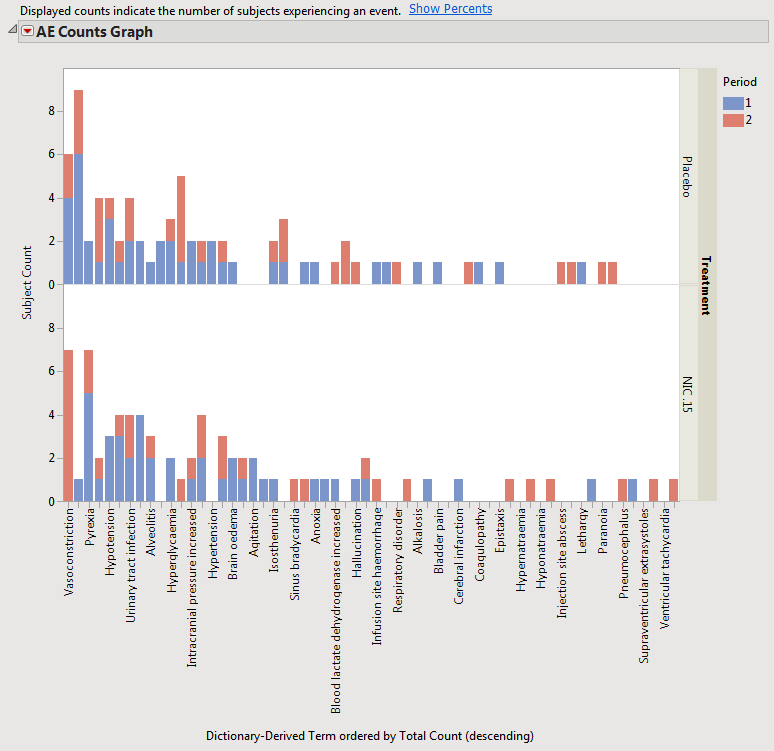
This view is similar for Interventions and other Events domains for distributions.