Note
: JMP Clinical employs a special protocol for data including non-unique Findings test names. Refer to
How does JMP Clinical handle non-unique Findings test names?
for more information.
Running Findings Time to Event for
Nicardipine
generates the
Report
window shown below. Refer to the
Findings Time to Event
requirements description for more information.
Note
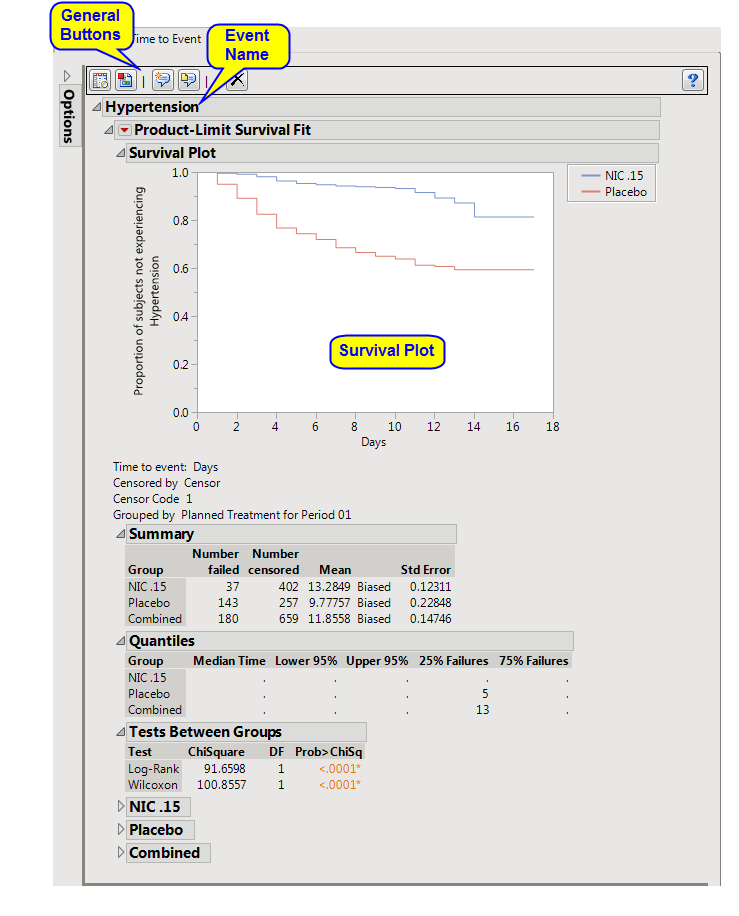
: In this example,
VS
was selected as the
Findings Domain to Analyze
,
Hypertension
was specified as the
Event Name
, and
SYSBP >= 140 and DIABP >= 90
was entered as the
Event Definition
.
|
•
|
Hypertension
: The name of this report corresponds to the
Event Name
supplied in the
dialog
. If this is
not
supplied, it is called
Domain Event
where
Domain
is determined by the
Findings Domain to Analyze
selected in the dialog. For example, if the event is defined using the
VS
domain, the tab is called
VS Event
. This tab contains a
Kaplan-Meier
analysis of the time to the defined event.
|
This analysis compares the time until the event among treatments in the study. Subjects
not
experiencing the defined event are
censored
at the date of their last available findings data.
See
Survival Plot
for more information.
|
•
|
Click
|
|
•
|
Click
|
|
•
|
Click
|
|
•
|
Click
|
|
•
|
Click the
arrow to reopen the completed process dialog used to generate this output.
|