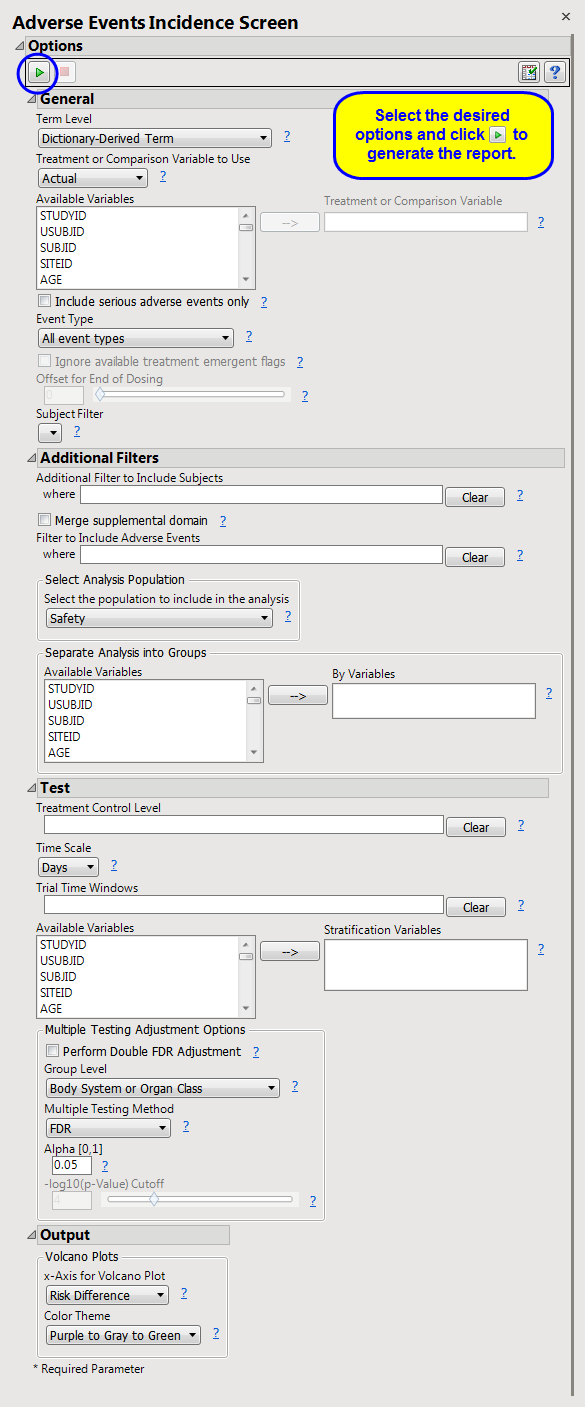
The
AE
Incidence Screen
report screens all
adverse events
by performing a
Cochran-Mantel-Haenszel exact test
on all 2 x 2 tables constructed from event incidence and treatment
arm
. Output one or more
volcano plots
of risk differences when using default settings. However, plots of relative risks or odds ratios can be generated depending on the selected option for the
X-Axis for Volcano Plot
. If
Study ID
varies among the subjects for analysis, the test is stratified by
Study ID
.
|
•
|
Perform Double FDR Adjustment
checked,
|
|
•
|
Group Level
set to
Body System or Organ Class
, and
|
|
•
|
Trial Time Windows
of
[1,6][7,12]
|
generates the report shown below. Refer to the
AE Incidence Screen
requirements description for more information.
The
Report
contains the following elements:
Presents a
Data Filter
and a
Volcano Plot
with the
adverse events
summarized at the selected
Group Level
. .
Note
: The name of this section reflects whatever term is selected as
Group Level
and is presented only if
Perform Double FDR Adjustment
is checked.
The
Body System or Organ Class Results
section contains the following elements:
|
•
|
This
Volcano Plot
displays
adverse events
summarized at the selected
Group Level
by
Trial Time Windows
(if specified). The
X
axis is chosen from
X-Axis for Volcano Plot
. In this example, the plot uses the difference in proportions between the treatments (
Risk Difference
). Other options include the
log
2
(Relative Risk
), which represents a doubling of the event rate for every one unit of change on the
X
axis, or the
log
2
(Odds Ratio
), which represents a doubling of the odds of an event for every one unit of change on the
X
axis.
In short, the smaller the
p
-value, the larger the number on the
Y
axis (
Y
can be thought of as the number of decimal places or number of zeros). Adverse events that are considered statistically significant while adjusting for multiple comparisons are above the dashed red line. This line is determined based on the selected
Multiple Testing Method
. The testing method considers the adverse event
Group Level
if
Perform Double FDR Adjustment
is checked.
p
-Value is from a
Cochran-Mantel-Haenszel exact test
. If
Study ID
varies among the subjects for analysis, the test is stratified by
Study ID
.
Bubble size
is an indicator of the total number of subjects experiencing the event. Because numerous adverse events could be represented at the
Group Level
, the most statistically significant individual term defined at the
Term Level
within each
Group Level
by
Trial Time s
(if specified) is presented on this tab. Because
Trial Time s
are defined, a
Group Level
bubble is presented separately for each time .
|
•
|
One
Data Filter
.
|
Note the following section-specific properties of the
Action Buttons
located at the top of the
Report
:
|
•
|
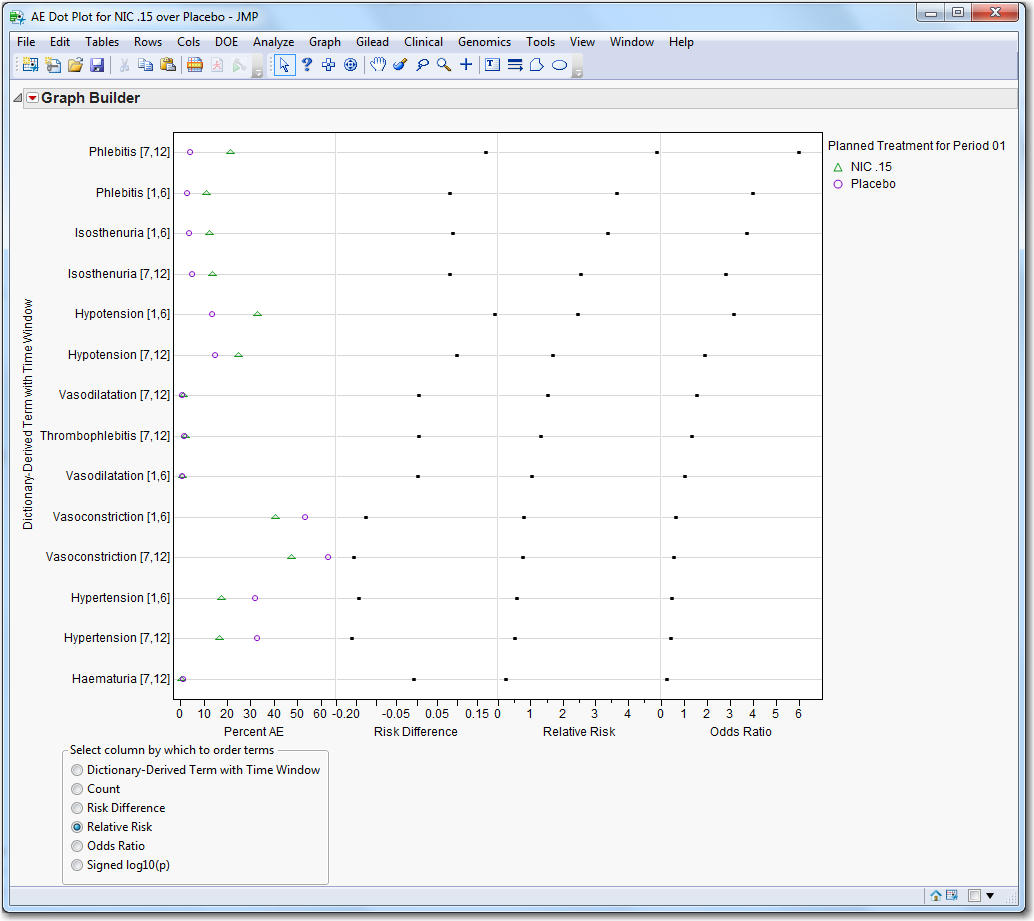
Dot Plot
: Click
|
|
•
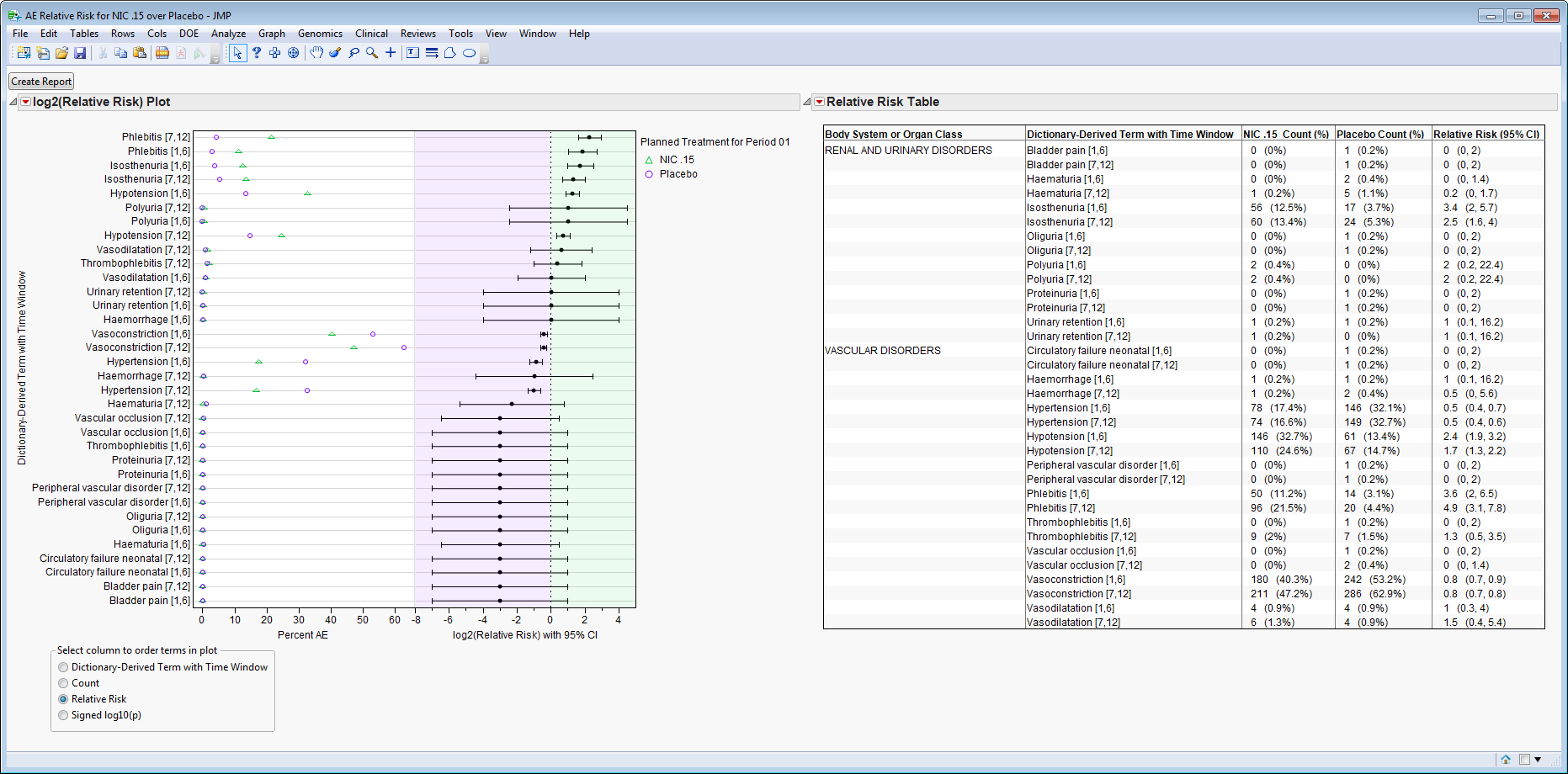
|
Relative Risk Plot
: Click
|
Relative Risk Table Example
: For the
Dictionary-Derived Term with Time
Phlebitis [7,12]
,
NIC .15 Count (%)
is listed as
96 (21.5%)
,
Placebo Count (%)
is listed as
20 (4.4%)
, and
Relative Risk (95% CI)
is listed as
4.9 (3.1, 7.8)
. This means that in Days 7 to 12 of the study, 96 subjects on the Nicardipine arm (which is 21.5% of all subjects on Nicardipine), and 20 subjects on the Placebo arm (which is 4.4% of all subjects on Placebo) experienced Phlebitis, with those in the Nicardipine arm 4.9 times more likely than those in the Placebo arm to experience it (with a 95% confidence interval between 3.1 and 7.8 more likely).
|
•
|
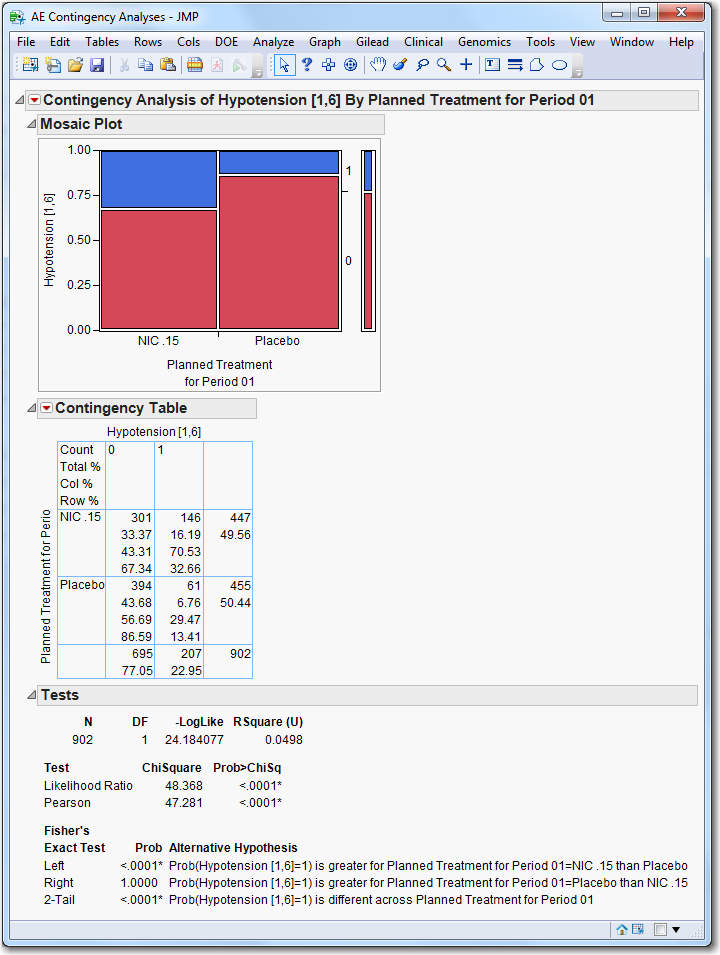
Contingency Analysis
: Click
|
|
•
|
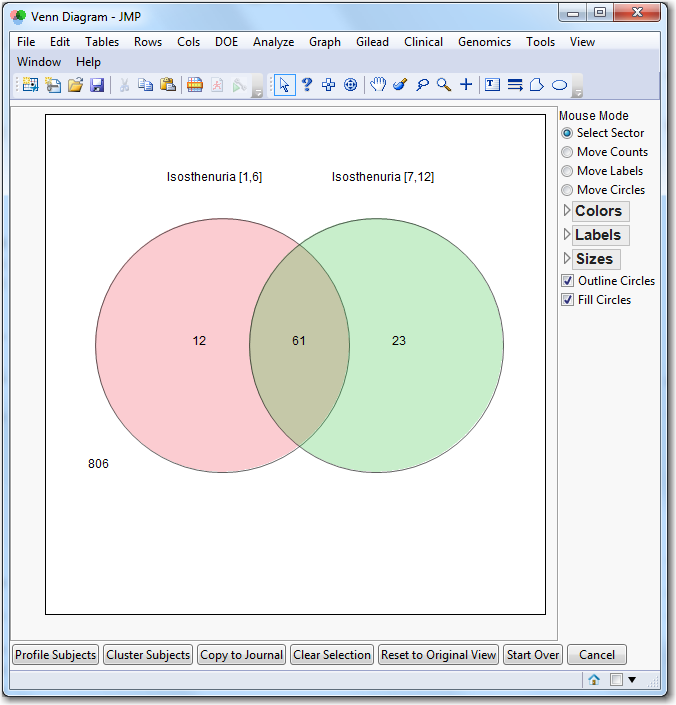
Venn Diagram
: Click
|
|
•
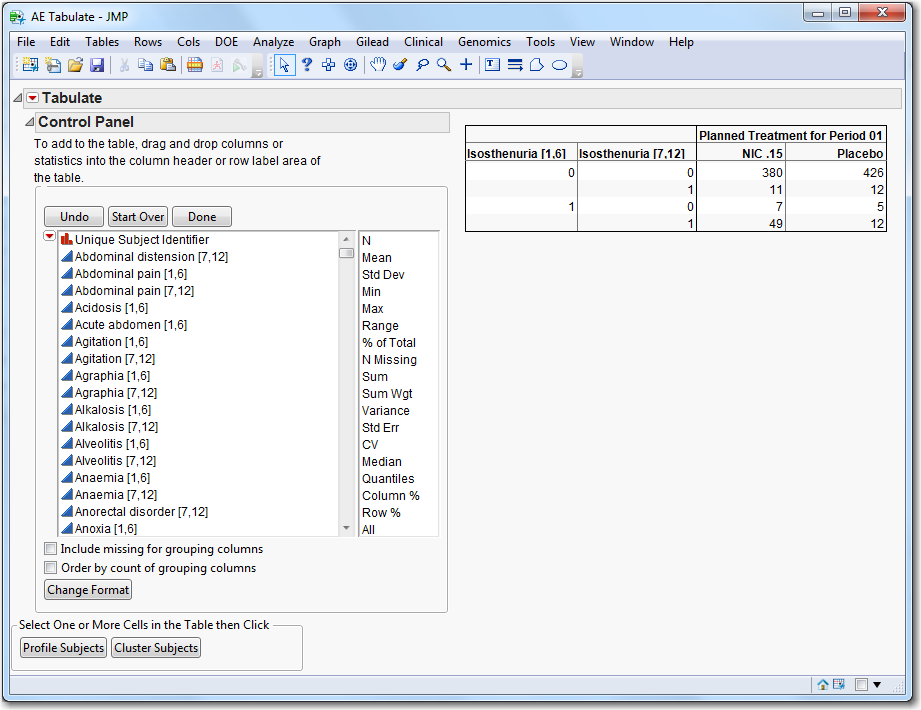
|
Tabulate
: Click
|
Contains a data filter and a
Bubble Plot
for each event defined at the
Term Level
for each statistically significant group and time (if present) that appears on the
Body System or Organ Class Results
section if
Perform Double FDR Adjustment
is checked. If
Perform Double FDR Adjustment
is
not
checked, all adverse events at the
Term Level
are presented. Note that the name of this section reflects whatever term is selected as
Term Level
.
Note
: The name of this section reflects whatever term is selected as
Term Level
. If
Perform Double FDR Adjustment
is checked, the terms for each statistically significant group and time (if present) that appear on the
Body System or Organ Class Results
section are presented. If
Perform Double FDR Adjustment
is
not
checked, all
adverse events
at the
Term Level
are presented.
The
Dictionary-Derived Term Results
section contains the following elements:
|
•
|
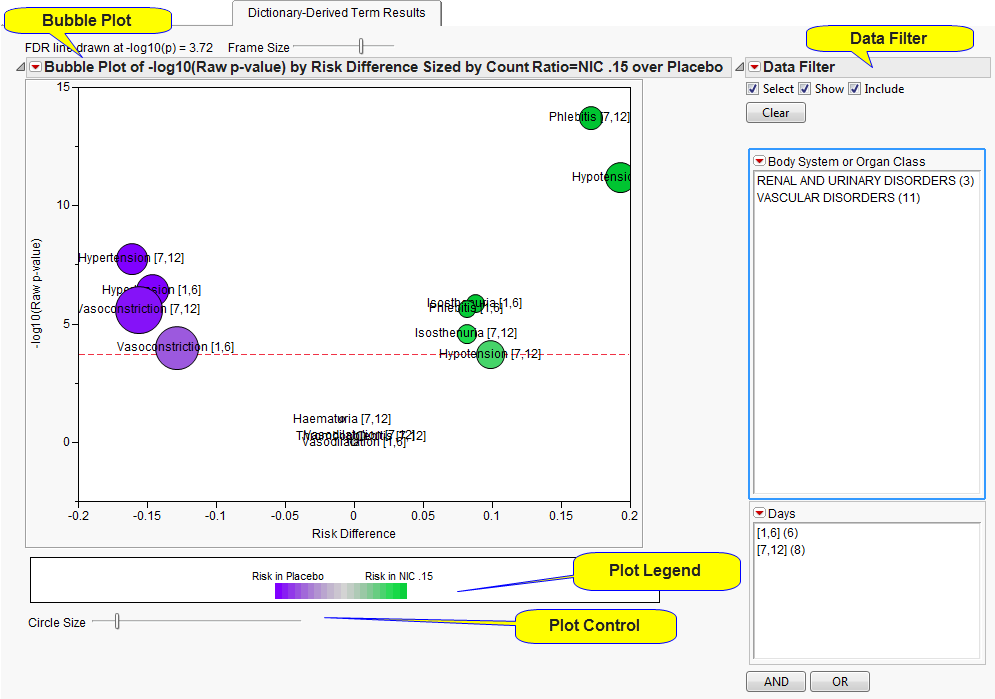
This
Volcano Plot
displays adverse events summarized at the selected
Group Level
by
Trial Time Windows
(if specified). The
X
axis is chosen from
X-Axis for Volcano Plot
. In this example, the plot uses the difference in proportions between the treatments (
Risk Difference
). Other options include the
log
2
(Relative Risk
), which represents a doubling of the event rate for every one unit of change on the
X
axis, or the
log
2
(Odds Ratio
), which represents a doubling of the odds of an event for every one unit of change on the
X
axis.
In short, the smaller the
p
-value, the larger the number on the
Y
axis (
y
can be thought of as the number of decimal places or number of zeros). Adverse events that are considered statistically significant while adjusting for multiple comparisons are above the dashed red line. This line is determined based on the selected
Multiple Testing Method
. The testing method considers the adverse event
Group Level
if
Perform Double FDR Adjustment
is checked. The
p
-value is from a
Cochran-Mantel-Haenszel exact test
. If
Study ID
varies among the subjects for analysis, the test is stratified by
Study ID
.
Bubble size
is an indicator of the total number of subjects experiencing the event. Because numerous adverse events could be represented at the
Group Level
, the most statistically significant individual term defined at the
Term Level
within each
Group Level
by
Trial Time s
(if specified) is presented on this tab. Because
Trial Time s
are defined, a
Group Level
bubble is presented separately for each time .
|
•
|
One
Data Filter
.
|
Contains a data filter and an animation across the time s of the results included on the
Body System or Organ Class Results
section.
Note
: The name of this section reflects whatever term is selected as
Group Level
and is presented only if
Trial Time Windows
are provided and
Perform Double FDR Adjustment
is selected.
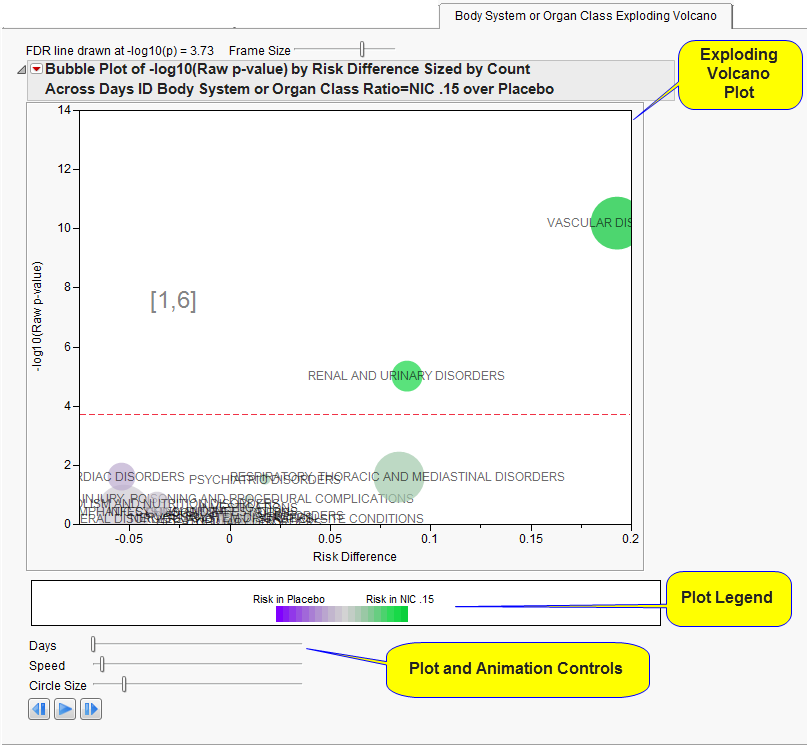
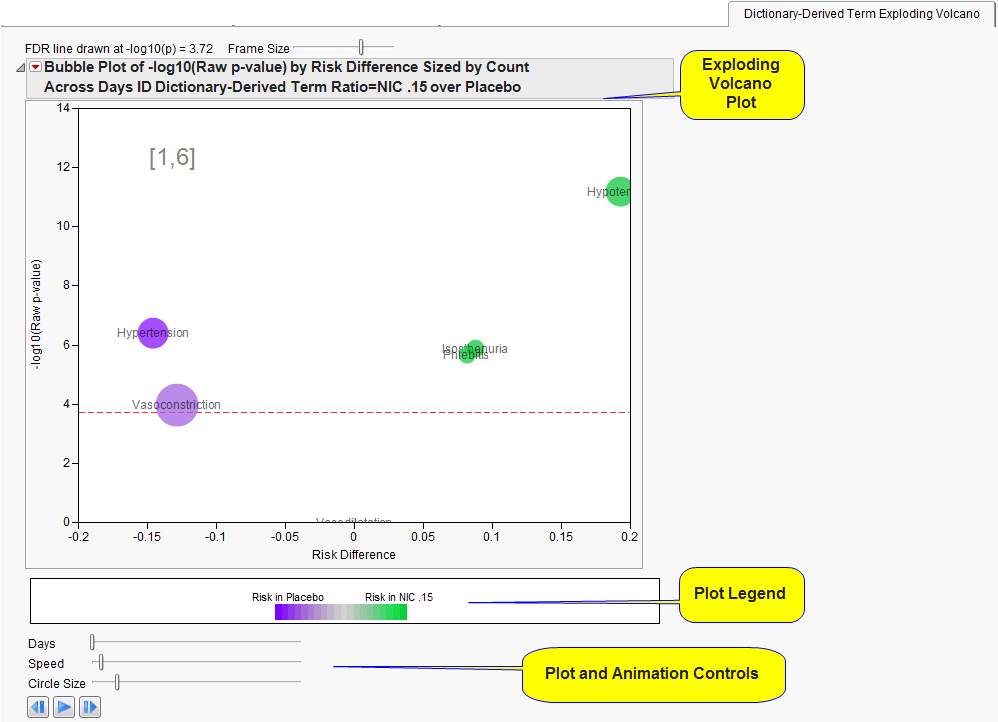
The
Body System or Organ Class Exploding Volcano
section contains the following elements:
|
•
|
This figure is animated and shows how the incidence of
adverse events
between the treatments changes over the course of the specified
Trial Time Windows
.
|
•
|
|
•
|
|
•
|
Contains a data filter and an animation across the time s of the results included on the
Dictionary-Derived Term Results
tab, which presents a separate bubble for each term or time .
Note
: The name of this section reflects whatever term is selected as
Term Level
and is presented only if
Trial Time Windows
are provided.
The
Dictionary-Derived Term Exploding Volcano
section contains the following elements:
|
•
|
This figure is animated and shows how the incidence of
adverse events
between the treatments changes over the course of the specified
Trial Time Windows
.
|
•
|
|
•
|
|
•
|
|
•
|
Dot Plot
: Click
|
|
•
|
Relative Risk Plot
: Click
|
|
•
|
Odds Ratio Plot
: Click
|
|
•
|
Contingency Analysis
: Click
|
|
•
|
Venn Diagram
: Click
|
|
•
|
Tabulate
: Click
|
|
•
|
Click
|
|
•
|
Click
|
|
•
|
Click
|
|
•
|
Click
|
|
•
|
Click the
arrow to reopen the completed report dialog used to generate this output.
|
|
•
|
Click the gray border to the left of the
Options
tab to open a dynamic report navigator that lists all of the reports in the review. Refer to
Report Navigator
for more information.
|
Note
: For information about how treatment emergent adverse events (TEAEs) are defined in JMP Clinical, please refer to
How does JMP Clinical determine whether an Event Is a Treatment Emergent Adverse Event?
.
Subject-specific filters must be created using the
Create Subject Filter
report prior to your analysis.
For more information about how to specify a filter using this option, see
The SAS WHERE Expression
.